Ecotest
ECOTEST - Saliva Pen Tests
ECOTEST - Saliva Pen Tests
Couldn't load pickup availability
Bulk Orders: info@ozmedical.com.au
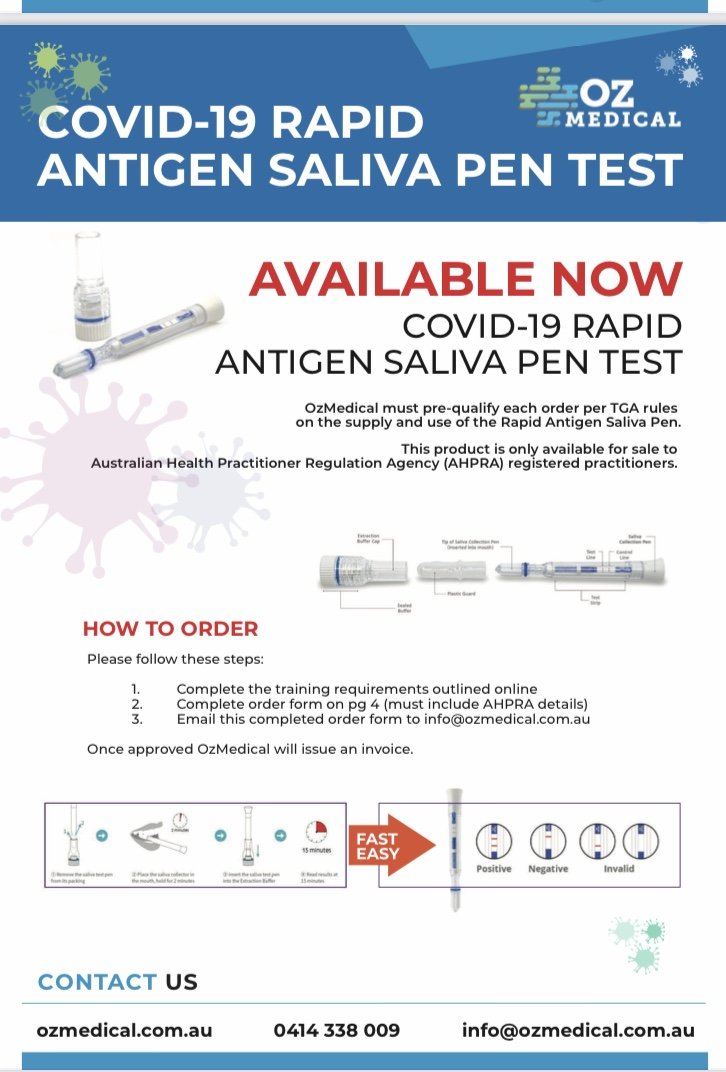
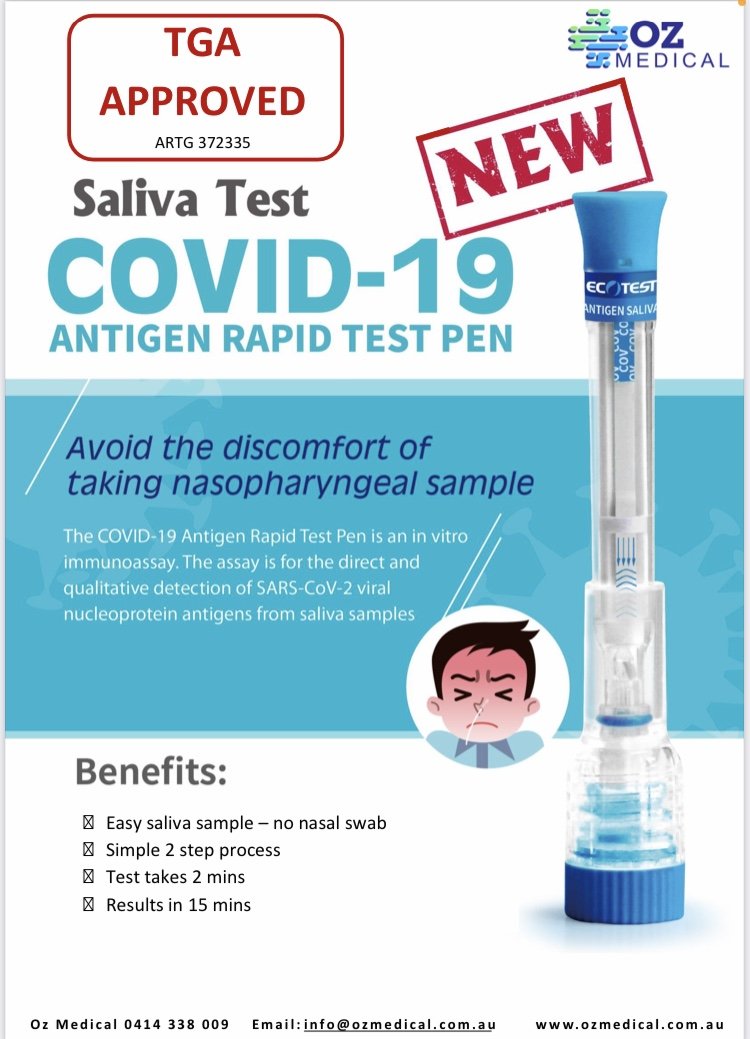
The COVID-19 Rapid Antigen Saliva Pen is an easy-to-use non-invasive technology enabling rapid testing anywhere, anytime.
3 Easy Steps to purchase :
1) - Simply buy now
2) - We will send you a Pre-Qualification consent form & a link to the training video
3) - Once you have watched the video we will dispatch your order
OZMEDICAL is pleased to be a major distribution partner - for the innovative ECOTEST Saliva Pen Test -In boxes of 20
Antigen Pen Training Record Template
Antigen Pen Testing Record Template
Antigen Pen Eco Test Instructions
PRODUCT NAME - ECOTEST
COVID-19 Rapid Antigen Saliva Pen Test (ECOTEST)
STANDARDS ACHIEVED
Product name: Severe acute respiratory syndrome-associated coronavirus IVDs
Sponsor: Emergence Technology Pty Ltd
Manufacturer: Assure Tech (Hangzhou) Co Ltd
Saliva test Expire July 2024- FROM TGA WEBSITE
| Name of self-test* and how to use the test | Sample type used | Australian Sponsor (supplier) | Manufacturer | ARTG | Clinical Sensitivity | Date Approved | Shelf Life |
|---|---|---|---|---|---|---|---|
|
Ecotest COVID-19 Antigen Saliva Test kit (COV-S35Pen) (pdf,447kb) (Rapid antigen test) |
Saliva | Emergence Technology Pty Ltd | Assure Tech (Hangzhou) Co Ltd (China) | 372335 | Acceptable sensitivity | 22 October 2021 | 33 Months (extended) |
|
Ecotest COVID-19 Antigen Nasal Test Self Test (pdf,451kb) (Rapid antigen test) |
Nasal swab | Emergence Technology Pty Ltd | Assure Tech (Hangzhou) Co Ltd (China) | 372335 | Very high sensitivity | 5 May 2022 | 30 Months (extended) |
NASAL Test Expire NOVEMBER 2024
PRODUCT NAME
COVID-19 Rapid Antigen Saliva Pen Test (EcoTest)
STANDARDS ACHIEVED
Product name: Severe acute respiratory syndrome-associated coronavirus IVDs
Sponsor: Emergence Technology Pty Ltd
Manufacturer: Assure Tech (Hangzhou) Co Ltd
TGA Search -- 372335 (funnelback.com) - ARTG ID 372335
Product name: Severe acute respiratory syndrome-associated coronavirus IVDs
Sponsor: Emergence Technology Pty Ltd
Manufacturer: Assure Tech (Hangzhou) Co Ltd
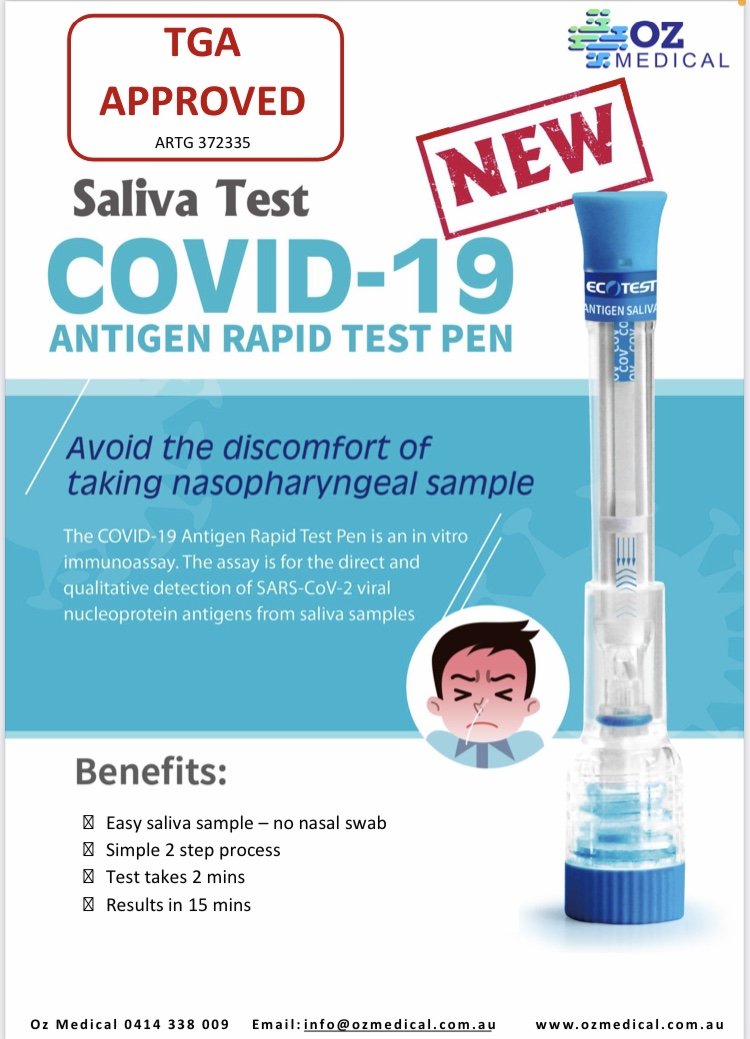
KEY FEATURES
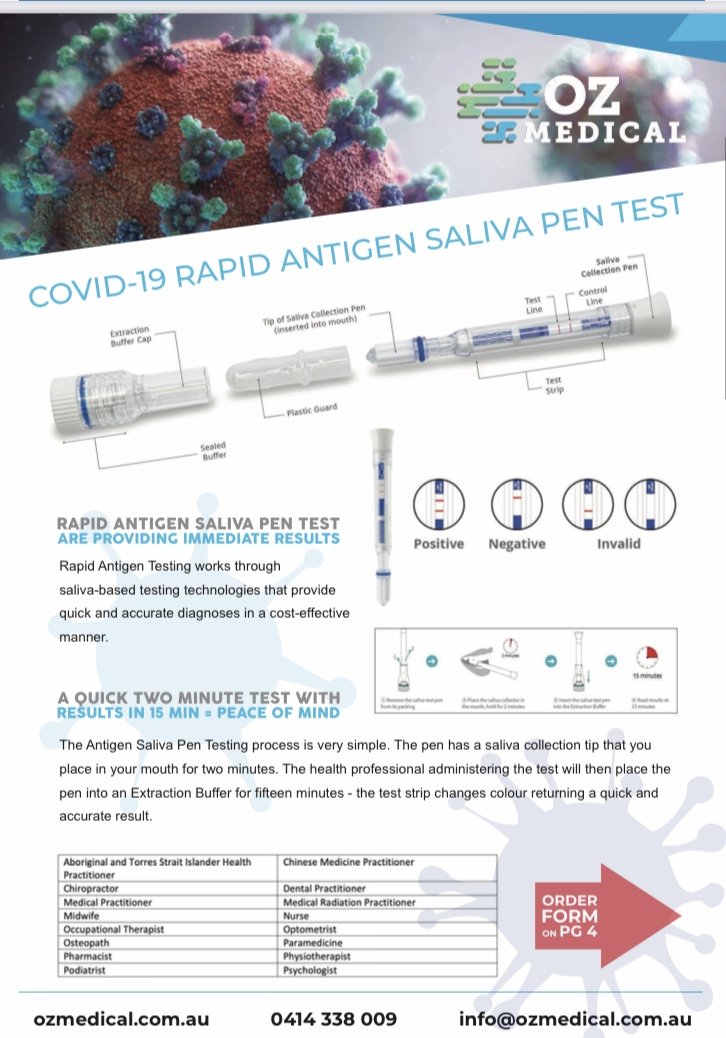
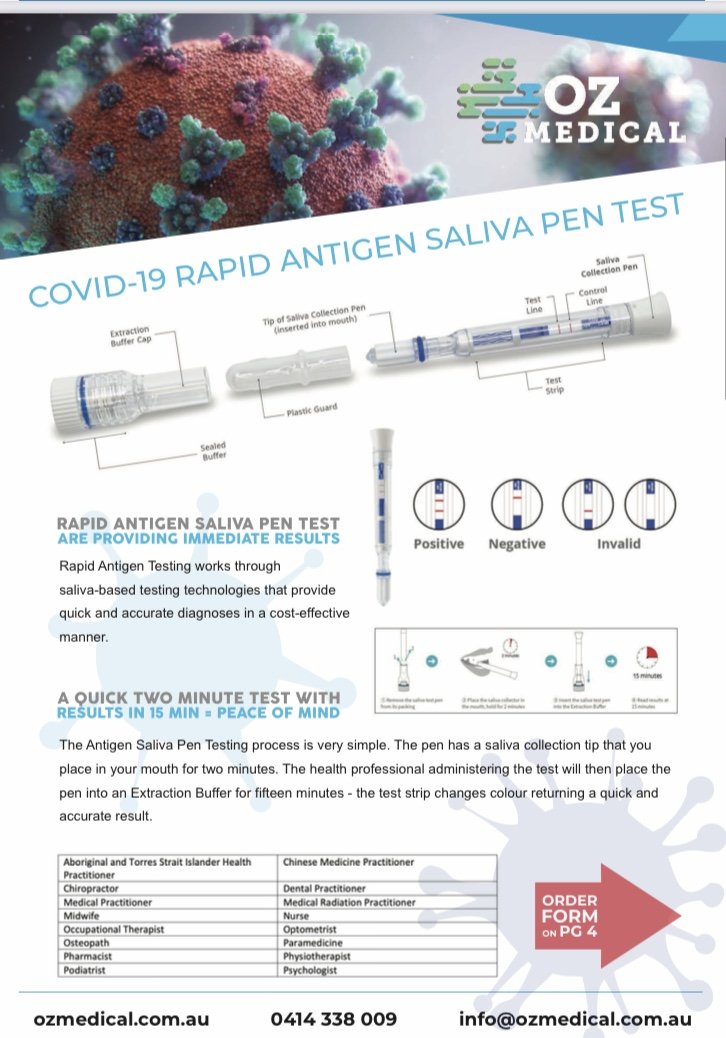
The COVID-19 Rapid Antigen Saliva Pen is an easy-to-use non-invasive technology enabling rapid testing with supervision by a health professional.
Results can be achieved in 15 minutes.
Negative test results do not exclude infection of COVID-19 (face masks, social distancing and good hygiene practices must be maintained).
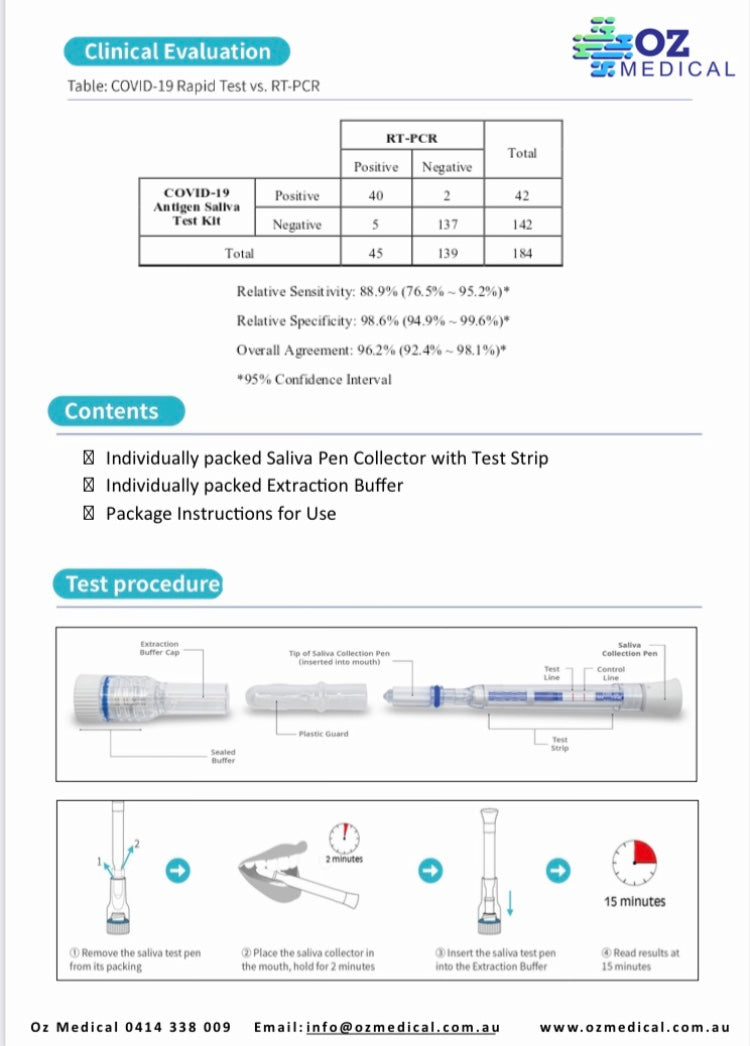
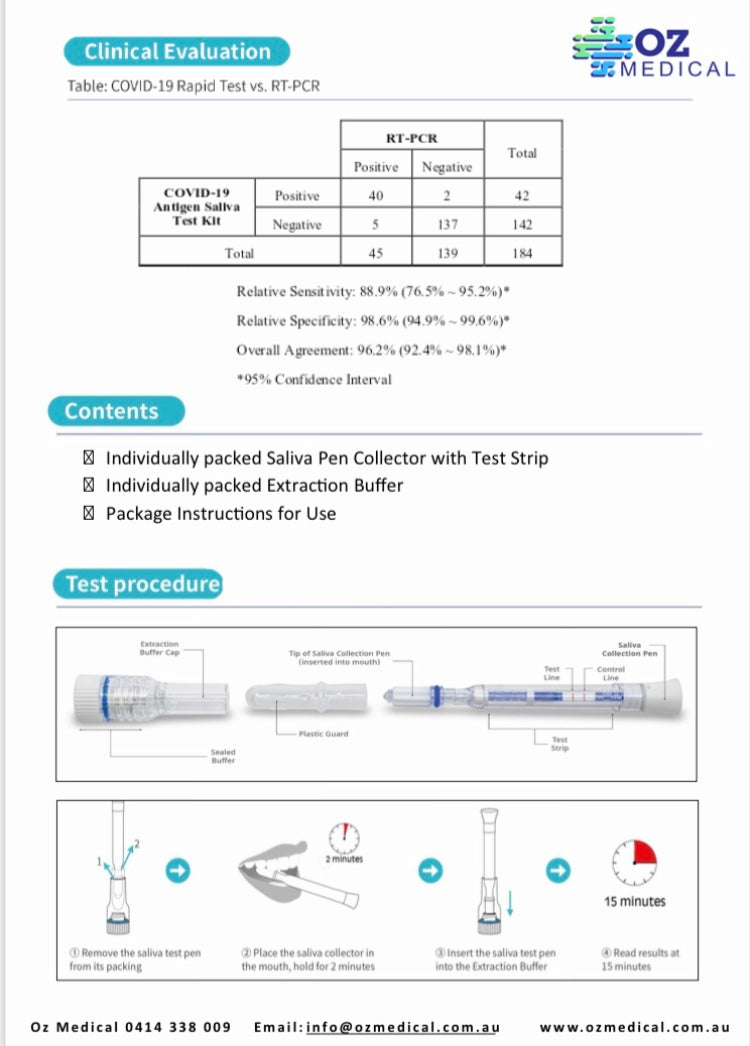
- Relative sensitivity: 90.0%
- Relative specificity: 97.0%
- Overall agreement: 95.3%
OzMedical must pre-qualify each order per TGA rules on the supply and use of the Rapid Antigen Saliva Pen.
OzMedical can only sell rapid test products to health practitioners or organisations who engage a health practitioner, who have received training in how to perform the test per TGA rules on the supply and use of the Rapid Antigen Devices.
Key training points:
-
Rapid Tests can only be performed by a trained health practitioner
Health Practitioner as defined by the Therapeutic Goods Act 1989 means: a person who, under a law of a State or internal Territory, is registered or licensed to practice in any of the following health professions:
-
Aboriginal and Torres Strait Islander health practice
-
dental (not including the professions of dental therapist, dental hygienist, dental prosthetist or oral health therapist)
-
medical
-
medical radiation practice
-
nursing
-
midwifery
-
occupational therapy
-
optometry
-
pharmacy
-
physiotherapy
-
podiatry
-
psychology
The list above includes medical practitioners, pharmacists, and nurses along with others, but not for example pharmacy assistants, dental assistants, or personal care workers in aged care.
Paramedics are not included in the definition of Health Practitioner in the Act but have been specified as a suitable health practitioner for the purposes of supply and use of rapid antigen tests.
-
-
Rapid tests can also be performed by a person trained by a health practitioner and then supervised by a health professional
-
The test or test training must be recorded and signed by supervising health practitioner
-
A health practitioner must review the interpretation of the results
-
If test is positive then contact National Covid hotline and person must have a PCR test.
A medical Health Practitioner must be engaged to perform or supervise the tests
Please phone Lisa on 0431 000 991 or email us anytime on info@ozmedical.com.au
Bulk orders are all available
20 units in a Box
www.skynews.com.au/australia-news/coronavirus/tga-approves-covid19-rapid-antigen-test/news-story/0ce4c23fe2ff064905f3d9375a892023
Currently rapid test products can only be sold to health practitioners or organisations who engage a health practitioner
For more information Contact Us.
Share